Oxalyl Chloride
Other Names:
Ethanedioyl dichloride
General Information:
Structure:
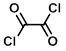
CAS Number: 79-37-8
Molecular Weight: 126.93 g/mol
Appearance: Colorless liquid
Melting Point: -12 C
Boiling Point: 63-64 C
Density: 1.48 g/mL
Common Uses:
Reagent for the conversion of carboxylic acids to acid chlorides
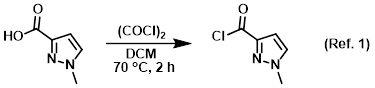
Procedure excerpt:
To a suspension of the SM (0.3 g, 2.38 mmol) in DCM (10 mL) was added (COCl)2 (0.3 mL, 3.6 mmol) dropwise at 0 C. The resulting mixture was stirred at RT for 1 h then . . .
Reagent in the conversion of primary alcohols to adehydes via Swern oxidation
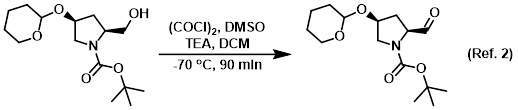
Procedure excerpt:
To a solution of (COCl)2 (19.0 g, 0.15 mol) in dry DCM (180 mL) was added dropwise a solution of dry DMSO (22.0 g, 0.30 mol) in dry DCM (60 mL) at -70 C under N2. The mixture . . .
Reagent in the conversion of secondary alcohols to ketones via Swern oxidation
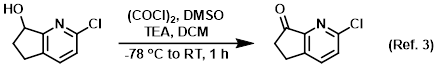
Procedure excerpt:
. . . solution of (COCl)2 (0.49 mL, 5.5 mmol) in DCM (12 mL) was slowly added DMSO (0.79 mL, 11.0 mmol) at -78 C. The resulting mixture was stirred at -78 C for . . .
Safety:
Oxalyl chloride is very toxic and corrosive. It is severely irritating to eyes, skin, and the respiratory tract. Oxalyl chloride reacts violently with water.
References:
1) Patent Reference: WO2010038081, page 138, ![]() (33.8 MB)
(33.8 MB)
2) Patent Reference: WO2010016005, page 120, ![]() (11.3 MB)
(11.3 MB)
3) Patent Reference: WO2016011390, page 112, ![]() (20.2 MB)
(20.2 MB)
4) Wikipedia: Oxalyl chloride (link)
5) www.sigmaaldrich.com: Oxalyl chloride (link)
6) Pearson, A. J.; Roush, W. R.; Handbook of Reagents for Organic Synthesis; Activating Agents and Protecting Groups